What is a ventricular assist device?
A ventricular assist device (VAD), is a mechanical circulatory device that is used to partially or completely replace the function of a failing heart. Some VADs are intended for short term use, typically for Patients recovering from a Heart Attack(Acute Myocardial Infarction) or heart surgery(Coronary Artery Bypass Graft (CABG), while others are intended for long term use (months to years and in some cases for life), typically for Patients suffering from congestive Heart Failure.
VADs need to be clearly distinguished from artificial hearts, which are designed to completely take over cardiac function and generally require the removal of the Patient’s heart.
VADs are designed to assist either the right (RVAD) or left (LVAD) ventricle, or both at once (BiVAD). Which of these types is used depends primarily on the underlying heart disease and the pulmonary arterial resistance that determines the load on the right ventricle.
LVADs are most commonly used, but when pulmonary arterial resistance is high, right ventricular assistance becomes necessary. Long term VADs are normally used to keep Patients alive with a good quality of life while they wait for a Heart Transplant(known as a “bridge to transplantation”). However, LVADs are sometimes used as destination therapy and sometimes as a bridge to recovery. Sometimes LVAD’s are used after ECMO(Extracorporeal Membrane Oxygenation) has been used and weaned.
In the last few years, VADs have improved significantly in terms of providing survival and quality of life among recipients.
Ventricular Assist Device image
How Does It Work?
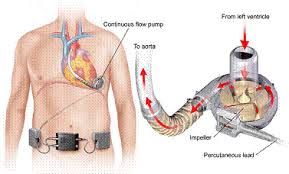
The device will be put in place by heart surgeons and is commonly in 3 sections. There is a single use blood pump, tubing to connect the pump to the heart’s blood vessels and a console that controls the VAD itself. The tubing is placed in the left chamber of the heart that pumps blood out to the body. Blood is pulled form the ventricle into the pump. The pump then sends the blood into the aorta, the large blood vessel leaving the ventricle.
The pumps used in VADs can be divided into two main categories – pulsatile pumps, that mimic the natural pulsing action of the heart, and continuous flow pumps. The pumps are commonly inserted by heart(cardiac) surgeons in the operating theatre.
Pulsatile VADs use positive displacement pumps. In some of these pumps, the volume occupied by blood varies during the pumping cycle, and if the pump is contained inside the body then a vent tube to the outside air is required.
Continuous flow VADs normally use either centrifugal pumps or an axial flow pump. Both types have a central rotor containing permanent magnets. Controlled electric currents running through coils contained in the pump housing apply forces to the magnets, which in turn cause the rotors to spin. In the centrifugal pumps, the rotors are shaped to accelerate the blood circumferential and thereby cause it to move toward the outer rim of the pump, whereas in the axial flow pumps the rotors are more or less cylindrical with blades that are helical, causing the blood to be accelerated in the direction of the rotor’s axis.
An important issue with continuous flow pumps is the method used to suspend the rotor. Early versions used solid bearings; however, newer pumps, some of which are approved for use in the EU, use either electromagnetic suspension (“maglev”) or hydrodynamic suspension. These pumps contain only one moving part.
Are There Any Complications?
Because the devices generally result in blood flowing over a non-biologic(pump) surface, predisposing the blood to clotting, there is need for anticoagulation measures. One device, the HeartMate XVE, is designed with a biologic surface derived from fibrin and does not require long term anticoagulation (except aspirin); unfortunately, this biologic surface may also predispose the patient to infection through selective reduction of certain types of leukocytes.
New VAD designs which are now approved for use in the European Community and are undergoing trials for FDA approval have all but eliminated mechanical failure.
VAD-related infection can be caused by a large number of different organisms:
- Gram positive bacteria (Staphylococci, especially Staph. aureus, Enterococci)
- Gram negative bacteria (Pseudomonas aeruginosa, Enterobacter species, Klebsiella species)
- Fungi. especially Candida species
Treatment of VAD-related infection is exceedingly difficult and some Patients die of infection despite optimal treatment. Initial treatment should be with broad spectrum antibiotics, but every effort must be made to obtain appropriate samples for culture. A final decision regarding antibiotic therapy must be based on the results of microbiological cultures.
Other problems include immunosuppression(suppression of the immune system), clotting with resultant stroke, and bleeding secondary to anticoagulation. Some of the polyurethane components used in the devices cause the deletion of a subset of immune cells when blood comes in contact with them. This predisposes the patient to fungal and some viral infections necessitating appropriate prophylactic therapy.
Any Questions?
Of course, if you have any questions or concerns, please discuss them with the ICU nurses and doctors.
All Intensive Care interventions and procedures carry a degree of potential risk even when performed by skilled and experienced staff. Please discuss these issues with the medical and nursing staff who are caring for your loved one.
The information contained on this page is general in nature and therefore cannot reflect individual Patient variation. It is meant as a guideline to specific information which will be discussed with you by the Doctors and Nurses caring for your loved one. INTENSIVE CARE HOTLINE attests to the accuracy of the information contained here BUT takes no responsibility for how it may apply to an individual Patient. Please refer to the full disclaimer.
Related Articles:
- How long does a cardiac arrest patient stay in Intensive Care?
- Six weeks in Intensive Care after Cardiac Surgery and my mother isn’t getting any better
- How long can a breathing tube or an endotracheal tube can stay in?
- How long should a Patient be on a ventilator before having a Tracheostomy?
- How long is a Patient kept on a BIPAP machine in Intensive Care?
- What is an induced coma and why is my critically ill loved one in an induced coma?
- The 3 most dangerous mistakes that you are making but you are unaware of, if your loved one is a critically ill Patient in Intensive Care
- How to always achieve your goals whilst your loved one is critically ill in Intensive Care
- Why you must make up your own mind about your critically ill loved one’s situation in Intensive Care even if you’re not a doctor or a nurse!
- Follow this proven 5 step process on how to be in control and influential if your loved one is a long-term Patient in Intensive Care
- How to quickly take control and have real power and influence if your loved one is critically ill in Intensive Care
- Why does my loved one need a Tracheostomy in Intensive Care?
- Tracheostomy and weaning off the ventilator in Intensive Care, how long can it take?
- My sister has been in ICU for 21 weeks with Tracheostomy and still ventilated. What do we need to do?
- Severe lung failure and my aunty is not expected to survive…
- 3 quick steps on how to position and prepare yourself well mentally, whilst your loved one is critically ill in Intensive Care
- How to get what you want whilst your loved one is critically ill in Intensive Care
- 5 steps to become a better negotiator if your loved one is critically ill in Intensive Care
- How to make sure that your values and beliefs are known whilst your loved one is critically ill in Intensive Care
- How to make sure that “what you see is always what you get” whilst your loved one is critically ill in Intensive Care
- What the doctors and the nurses behaviour in Intensive Care is telling you about the culture in a unit
- How long does it take to wake up from a Traumatic brain injury or severe head injury
- How to take control if your loved one has a severe brain injury and is critically ill in Intensive Care
- Family Meetings in Intensive Care or the Elephant in the Room
- What you need to do if your loved one is dying in Intensive Care(part one)
- What you need to do if your loved one is dying in Intensive Care(part two)
- Intensive Care at its best?
- How INTENSIVECAREHOTLINE.COM Can Help You
- What you and your Family need to do if your critically ill loved one is very sick in Intensive Care and faces an uncertain future
- How long can somebody stay in Intensive Care?
- My Family can’t agree on what’s best for my sister in Intensive Care…Help!
- My husband is dying in Intensive Care, but we need more time…
- My mother sustained serious brain damage after a stroke and she now is in multi- organ failure